Medical Research That Reaches Outer Space
Scientists seeking to cure a rare condition may have found a way to help astronauts

One of NASA’s missions is to see the first humans set foot on Mars, but microgravity – which causes astronauts to float in space – can have significant to severe physiological effects. Medical researchers at the University of Birmingham looking for ways to alleviate brain pressure have found a drug that may also be suitable for voyaging to outer space.
In the past when astronauts traveled to the moon, their trips lasted a mere three days. But a voyage to Mars is a much lengthier venture, up to nine months in transit in microgravity prior to setting foot on the surface. As space agencies set their eyes on more ambitious targets, their journeys will take a toll on astronauts’ bodies and make exposure to risks more likely.
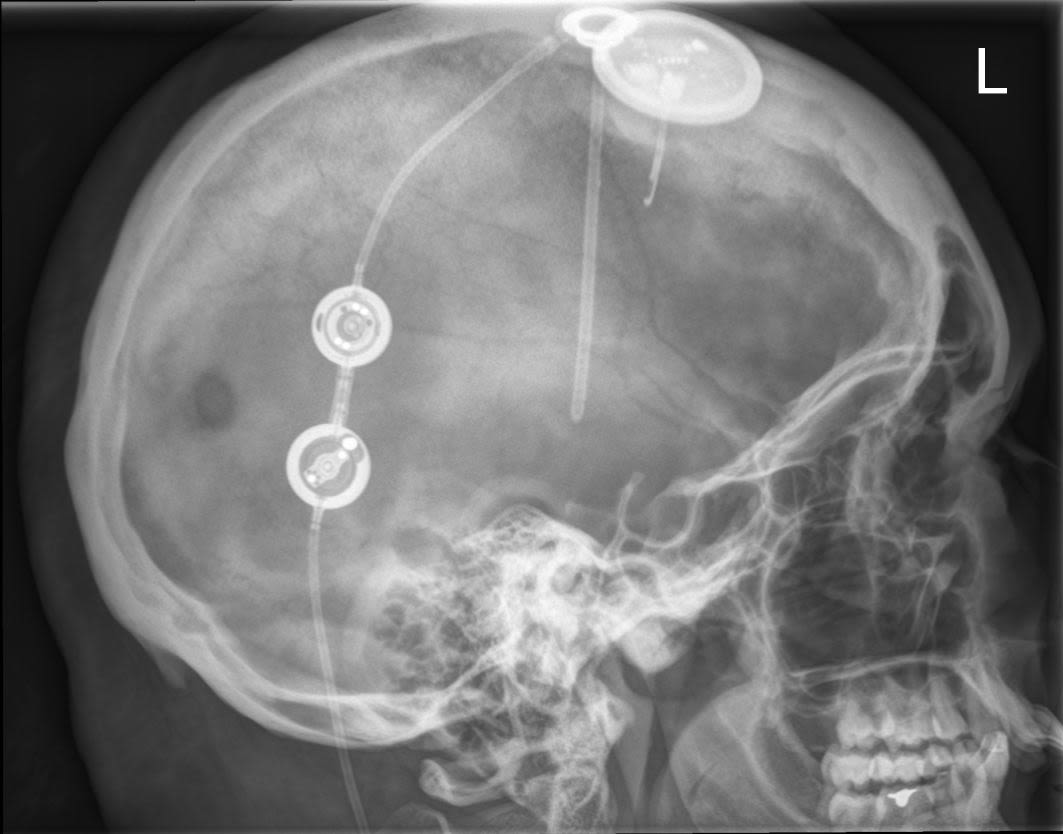
Skull x-ray of a patient with Idiopthic Intracranial Hypertnesion showing a tube (shunt) and pressure monitor in place for a patient who was developing blindness and needed emergency surgery to rapidly lower her intracranial pressure to save vision.
Skull x-ray of a patient with Idiopthic Intracranial Hypertnesion showing a tube (shunt) and pressure monitor in place for a patient who was developing blindness and needed emergency surgery to rapidly lower her intracranial pressure to save vision.
Professor Alexandra Sinclair is a neurologist and academic who has spent years researching idiopathic intracranial pressure (IIH), a condition with similar effects on the body as the brain pressure caused by space travel. A key aim of her work at the University of Birmingham’s Institute of Metabolism and Systems Research is to translate discoveries from her research into practice that benefits patients directly.
Although its cause is unknown, the impact of IIH is inescapable for sufferers. They endure severe headaches that can last for decades, and risk losing their vision. Treatments are limited to invasive surgery, lifestyle changes, or medications largely viewed as inadequate. Now, after decades of little progress, a new drug is on the horizon.
The findings, published by Sinclair and colleagues in Science Translational Medicine, could not only change the lives of IIH sufferers, but may also have applications for those interested in space travel. NASA’s medical team visited the University of Birmingham in June, to discuss intracranial pressure and how the new drug treatment might also be suitable for space travel.
A life-limiting condition
Sinclair, a trained neurologist, first encountered IIH as a junior doctor after being called to see a patient who had been rushed to hospital when she began to go blind. “I arrived on the ward to find a very frightened young, girl with obesity who had presented with difficulty seeing and they found that she had swelling at the back of the eye, which was papilledema, the characteristic of raised brain pressure,” she recalls.
A scan showed no tumor but by the time Sinclair saw her, the damage was significant, emergency neurosurgery was conducted to partially reverse the blindness. “This poor girl had lost almost all of her vision to the extent that she could only see my hands waving in front of her – she couldn’t even read down a chart,” Sinclair says. “I was really struck by a very young person going rapidly blind from this condition that was idiopathic intracranial hypertension.”
Thus began Sinclair’s interest in a condition that has puzzled clinicians and researchers alike since it was first recognized in the 1930s. A drug called acetazolamide, which was developed in the 1950’s, is typically given to patients, but doesn’t always work, and the side effects – tingling hands, a horrible taste in the mouth, and depression – make it difficult to tolerate. When blindness is an immediate risk, physicians undertake cerebrospinal fluid shunting surgery, inserting a tube from the ventricles of the brain into the abdominal cavity to release the brain pressure. But this doesn’t solve the problem and offers only temporary relief as the tubes (known as shunts) are prone to getting blocked or infected, and the procedure often needs to be redone.
IIH tends to affect a particular demographic – young, women with obesity. Alongside rising rates of obesity, rates of IIH doubled between 2002 and 20016, from 2 to 5 per 100,000 in the general population. By some definitions, it might no longer be considered “rare.”
The patients who take part in the Birmingham group’s research studies are referred by local opticians or hospitals. By the time they reach Sinclair’s lab they are understandably terrified, says Dr. James Mitchell, a clinical lecturer in neurology and former PhD student in Sinclair’s lab, who ushers them through the process. “They’ve started off with the news that they might have something they consider to be very serious [a tumor] — and usually a brain scan takes place to make sure that isn’t the case,” he says.
“It is extremely worrying and often people are very disabled by the headaches they’re having as well. The majority of people have no remission from the headaches, so it’s almost continuous,” affecting their lives and ability to work, he says.
Sinclair has led the first IIH management Guideline which has been key to improving patient care internationally.

In the laboratory where discovery science from Professor Sinclair’s lab is being translated into patient benefits with (from left to right) Dr James Polk, Chief Medical Officer of NASA, Mike Barratt astronaut who has completed two space walks, Professor Sinclair and Zerin Alimajstorovic postdoctoral researcher.
In the laboratory where discovery science from Professor Sinclair’s lab is being translated into patient benefits with (from left to right) Dr James Polk, Chief Medical Officer of NASA, Mike Barratt astronaut who has completed two space walks, Professor Sinclair and Zerin Alimajstorovic postdoctoral researcher.

Professor Alexandra Sinclair, University of Birmingham discussing raised intracranial pressure with Dr James Polk, Chief Medical Officer of NASA during the round table discussion held at University of Birmingham.
Professor Alexandra Sinclair, University of Birmingham discussing raised intracranial pressure with Dr James Polk, Chief Medical Officer of NASA during the round table discussion held at University of Birmingham.
New uses for a known drug
Sinclair was convinced patients needed better treatment options, and began to study secretary mechanisms, observing similarities between the brain and kidney. The kidney modulates fluid by a mechanism similar to the choroid plexus (though in reverse) — a group of cells responsible for secreting cerebrospinal fluid. Exenatide, a standard treatment for diabetes that modifies fluids in the kidney, might therefore have a similar effect in the brain. The hypothesis proved true: When the team conducted research in animals the results were highly promising. A trial in humans is now under way.
The fact that exenatide was already used for diabetes has allowed the research to advance quickly, bypassing the tests typically needed to prove a drug is safe for human consumption. “It’s going to be much quicker to bring this drug through for patients because we know that it is safe and what the side effect profile is. And we know that the doses that are being used for diabetes are similar to what we’re currently testing for raised brain pressure,” Sinclair says.
The research draws on multidisciplinary expertise spanning clinical neurology, medicine, ophthamology, and basic laboratory science. Zerin Alimajstorovic, who holds a PhD on causes of fluid secretion and drainage in the brain, was responsible for running experiments on mice, and deployed “spectral domain optical coherence tomography”—a sophisticated imaging technique to measure the retinal layers of the eye. This is also “a common tool used by the clinicians in our group on human patients when checking for signs of papilloedema or optical disc swelling as a means of monitoring IIH,” he explains. Optical coherence tomography is also a key technique used by NASA to monitor the ocular health of the astronauts. Alimajstorovic is currently in UCLA studying new methods for measuring intracranial pressure.
But raised brain pressure affects not just astronauts or people with IIH. When someone receives a blow to the head, the pressure in their brain will rise. Mitchell, who is also a doctor in the British Army, is hopeful that exenatide could be used in cases of traumatic brain injury to reduce the impact of swelling. It might also treat stroke, or help ameliorate a life-threatening condition called hydrocephalus – water retention in the brain – sometimes seen in babies.
This breadth of possible applications may be the most ground-breaking aspect of the discovery. “Potentially it has application to any condition where you see raised intracranial pressure,” Mitchell notes.
With the FDA and European Medicines Agency supporting orphan drug designation for this new use of exenatide, Sinclair is hopeful that it will be available to patients in the near future. In the near future, this seminal research could bring life-changing benefits both to adventurers in outer space and sufferers back here on earth.
For insight about our research visit: www.birmingham.ac.uk/quest


